A few weeks ago, the Lancet published an analysis which concluded that global mortality associated with antimicrobial resistance (AMR) has been greatly underestimated, and that with 3,500 daily deaths being directly related to AMR, it is one of the leading threats to human health worldwide. The analysis covers more than 200 countries and is the most comprehensive study on the consequences of antimicrobial resistance to date.
Tragically, people are dying from infections that previously were treatable. Children under the age of five are at particular risk in low- and middle-income countries (LMICs) – with more than a quarter of a million dying each year, or one child every two minutes.
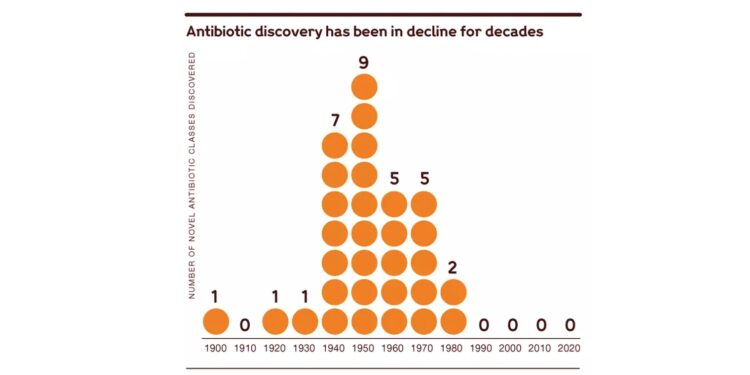
Decades of antibiotic overuse and misuse, in people and in animals, has caused the emergence of “superbugs”, strains of bacteria resistant to existing medicine, leaving the world vulnerable and decades of medical progress being undermined.
At the same time, the world has not seen the introduction of truly novel antibiotics for many years. Regrettably, an unsustainable innovation and payer ecosystem has caused the pipeline of drugs with new targets or mechanisms of action to dry up.
To be ahead of the curve, we need a robust antibiotics pipeline, delivering a sustainable supply of innovative new antibiotics over the coming decades.
Wellcome Trust and the Novo Nordisk Foundation have both been doing what we can to support vital antibiotic innovation, through our investments in CARB-X, the REPAIR Fund, and the AMR Action Fund. But while our investments have done much to shore up the early-stage pipeline, we now need governments to do their part and ensure that antibiotics can reach global markets – available, accessible, and affordable. With this in mind, we recently commissioned Boston Consulting Group and the World Economic Forum to produce a report with recommendations on how to address these remaining gaps in support for antibiotic R&D. The report provides a roadmap for global leaders to study and adopt.
The effects of a broken antibiotics market
The report concludes that we need new incentive structures that will enable both biotech and pharmaceutical companies to invest in the development and commercialization of new antibiotics.
Over the last years, we have seen biotech companies going bankrupt and pharmaceutical companies withdrawing from investing in the development of new antibiotics. What is perhaps even worse, we see scientists – crucial to delivering the new innovation we need – abandoning the cause.
We need to change the way we pay for antibiotics if we want to secure the introduction of novel treatments to save people.
A new antibiotic payment model
AMR policy must focus on advancing truly novel and innovative treatments by creating a sustainable innovation ecosystem that leverages existing private sector R&D and public-private collaborations. Crucially, governments must find a way to deliver a predictable revenue that is large enough to offset all the investment costs in R&D before the product is ready to enter the market.
The World Economic Forum and Boston Consulting Group have analysed five potential models for pull incentives, various versions of which have been piloted over time in relation to antibiotics as well as other treatment areas. The different methods have been tested on parameters delivering continuous innovation, portfolio diversity, access, appropriate stewardship, and supply availability.
The resulting recommendation is to apply a subscription payment model as the way forward. Such a payment scheme is already being piloted in the UK and Sweden. The subscription model is based on fixed annual payments for a set period, in return for a sufficient antimicrobial product supply guarantee, and, crucially, de-linked from the volumes sold. Recently, this has been dubbed the ‘Netflix model of antibiotics’ or compared to safeguarding properties by investing in fire extinguishers, hoping that there will never be a need for them.
The idea is not new, and political leaders have time and again made warm commitments to act to support antibiotic development. But now is the time to turn these into solid action and bring fresh political goodwill to get them implemented. In the US, the world’s largest pharmaceuticals market by some margin, legislators are currently considering such a subscription model. With the support of both political sides, the so-called PASTEUR ACT (The Pioneering Antimicrobial Subscriptions To End Up Surging Resistance) bill has been drawn up, precisely with the aim of creating more attractive conditions for the development of new antibiotics.
The subscription model is advantageous compared to the current alternatives, as it offers manufacturers security in terms of their revenues as well as certainty in terms of the demand, thereby providing financial predictability. In addition, a subscription model allows governments to reinforce proper stewardship and provides the certainty of fixed and predictable costs for years to come.
The conclusion of the report is that the subscription model – tailored to the needs and characteristics of individual countries – appears to be the most attractive and viable option for reinvigorating the AMR ecosystem and that it should be a serious consideration for all policy-makers. It addresses most of the requirements for rewarding innovation in a fair manner and ensures access and post-launch sustainability for new treatments. In this critical respect, it is superior to the other funding models analysed.
The solution requires political intervention to reconstruct the market and make the development, production of and marketing antibiotics profitable again. We need to change the way we value antibiotics —by paying for reliable and sustainable access to these lifesaving drugs.
The COVID-19 pandemic has highlighted the importance of global collaboration, having political leaders, the healthcare community, the private sector, and the public working together to tackle a global health threat. As with COVID-19, we know what needs to be done, and in order to succeed, now is the time for all stakeholders to join forces with a sense of urgency and global solidarity. If we fail, we have only seen the beginning of the human suffering and societal problems that antimicrobial resistance will entail.
(Courtesy World Economic Forum/By Jeremy Farrar, Mads Krogsgaard)